Digitization of processes and data across the value chain along with the emergence of Internet of Things (IoT) has transformed the Pharma industry. Although IoT is still in its nascent stages of adoption in the Life Sciences industry, the use of smart devices and machine-to-machine (M2M) communication leveraging SMAC technologies comes at a time when the industry is grappling with patent cliffs and declining R&D productivity.
The early signs of IoT adoption are quite palpable with some technology giants introducing patient centricity-based mobility products that provide real-time health monitoring and reporting, health care professionals (HCP) scheduling, medication adherence, etc.
Not only is IoT rapidly changing the patient experience, but also is making a dramatic difference in other areas of the industry such as R&D, clinical development and supply chain.
This paper talks of how IoT has the potential to transform the Life Sciences industry by offering value propositions such as improved patient and HCP engagement, cost optimization, faster time to market, higher productivity and improved compliance adherence.
Introduction
The Life Sciences industry had been more reactive than proactive in technology adoption, primarily because of tight regulations and domain complexities. But over the past few years, falling R&D productivity, increasing costs, compliance non-adherence, large number of patents expiring and increasing stakeholder expectations with respect to drug efficacy have put tremendous pressure on Pharma companies.
Some early adopters have already started exploring IoT to enable end-to-end digital integration across the value chain. IoT-based smart devices such as “Organ in a Chip,” which allows organizations to run real-life diagnostics scenarios, are already gaining traction. Clubbing the output from these devices with Big Data analytics and cognitive systems has the potential to provide unprecedented opportunity, thereby drastically improving hit rate and R&D productivity.
Another example of growing influence of smart devices is “Chip in a pill” - a special ingestible pill - that on consumption captures health status, including drug effects on key organs, and sends to a wearable device. This data is then sent as a report over cloud to HCP for diagnosis.
Use of smart devices in clinical development, supply chain and patient engagements can not only help reduce time-to-market for drugs but also the real-time data feeds can be ploughed back to proactively detect errors across the value chain and, thus, improve regulatory compliance. Data from wearable devices can be used by HCPs to prescribe personalized medicines (PM) that will improve drug efficacy manifold and will reduce treatment period.
Some of the fast growing IoT applications across the Pharma value chain are:
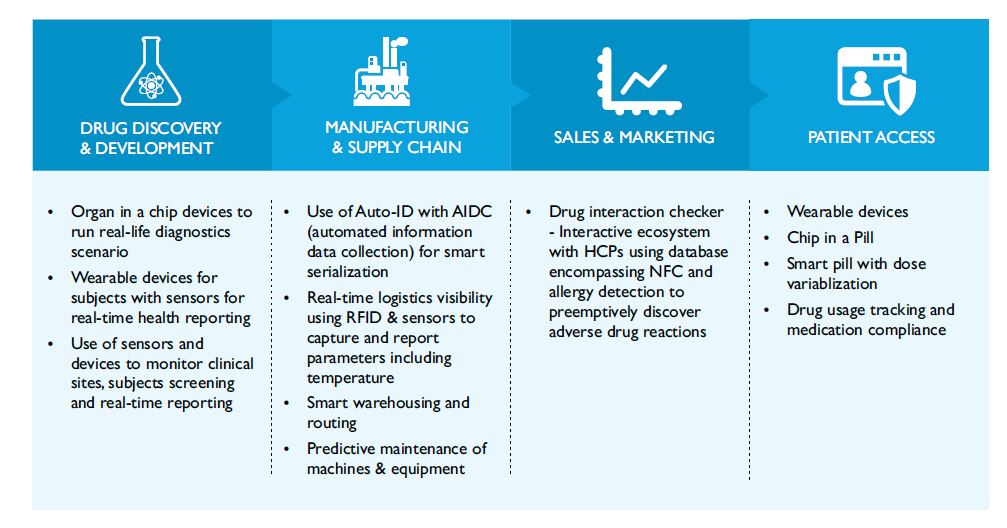
Figure 1: IoT Application Across Pharma Value Chain
Although IoT finds opportunities across the entire value chain, R&D, clinical development, supply chain and patient centricity are the areas where its intervention is likely to reap most benefits.
Need for an Architecture
Internet of Things requires companies to build and support a future-ready technology infrastructure. This architecture is made up of multiple layers, including new hardware, embedded software, connectivity, a cloud consisting of software running on remote servers, a suite of security tools, and integration with enterprise business systems and external environments.
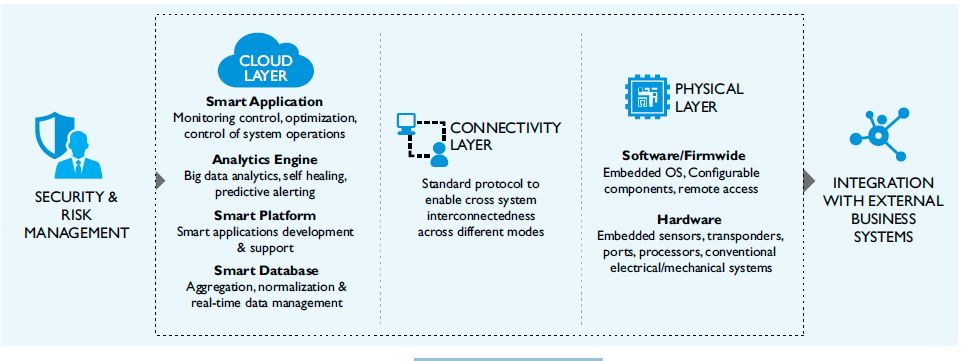
Figure 2: Internet of Things Architecture
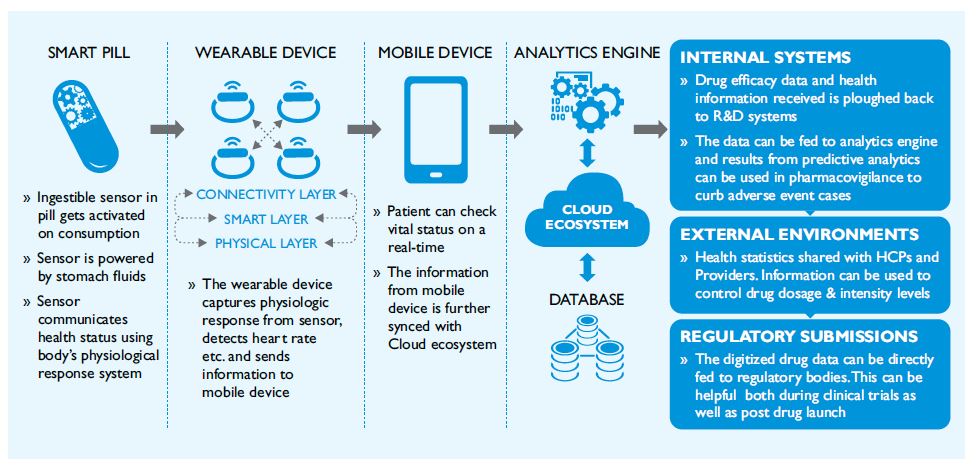
Figure 3: “Chip in a pill” Ecosystem leveraging IoT Architecture
Figure 3 shows how smart devices can capture health indicators of the wearer and compile, analyse and report insights to the cloud layer. The data is stored in the centralized database, analyzed with a bigger data set in the analytics engine to generate health insights. This information is categorized and shared using cloud layer with external and internal systems.
Weighing Pros and Cons
Implementation of an IoT ecosystem is not hassle free. It is imperative for a Pharma company to conduct a detailed due diligence to analyze the people, process and technology readiness for IoT. Key challenges that pharma companies need to deal with are:
IoT can help Pharma companies derive value and provide competitive advantage through:
Cost savings: This will be attained throughout the value chain
R&D: IoT applications such as “’Organ in a Chip” will drastically improve the productivity through rapid trials running multi-stage diagnostics. This will bring down the trial and data reporting costs and will also help reduce manual intervention in mundane activities. Advanced analytics tools can feed data directly to generate vital diagnostics and real-time reporting
Clinical development: Cost reduction will be achieved through savings attained in clinical trial phase as a result of continual feedback on subject diagnostics and monitoring using sensors, faster subject screening and analysis
Supply Chain: Reduction in operations and maintenance cost. Savings will also come from improved inventory planning reducing obsolescence cost and risk. The improved logistics planning will help in reducing redundant costs that was incurred due to wastages and incorrect supply. This will help bring down overall drug costs as well
Improved Quality: With smart systems, there will be a better control on precision and quality of drug produced and error rate would be reduced drastically. During the discovery and trial phases as well, smart reporting and analytical systems will help reduce adverse effect rate, thereby improving drug quality in terms of safety. The smart systems in patient access phase will help improve quality of care
Better compliance adherence: Smart devices will enable real-time data reporting to central systems. This will make the monitoring live and exceptions can be responded to quickly. This will help improve regulatory reporting, reducing manual intervention. Adverse event reporting can be minimized through predictive alerting
Better planning and lesser time to market: The real-time data reported across value chain components can be used for generating business value insights for leadership teams, thus resulting in near real-time decision making. The required feedback and changes related to drug research, efficacy, adoption, patient outcomes etc. can also be easily percolated back to the systems. This will be particularly useful during drug launch, drug pricing, and drug marketing related strategy development. Also, the efficiency improvement will optimize the time to market.
Conclusion
Internet of Things is a reality in today’s era of digitization and, therefore, it deems fit that Pharma companies adopt it at the earliest. Though IOT is still in its nascent stages of development and adoption across industries, it is imperative for Pharma companies to include IOT as part of their strategic focus. This will help them start exploring and implementing IOT applications across value chain components that are ailing and are potential candidates for IOT adoption. This would require companies to take reformative steps such as rehauling systems and processes and transforming business models using nextgen architectures.
On one hand, IoT offers added quality, agility and value to the business; on the other hand, it promises tremendous opportunities for innovation and can lead to a new era of transformation in Pharma.
Akash Shrivastava is a Lead Consultant in Wipro’s Life Sciences business.
He has over 7 years of experience in management consulting and presales roles and has worked with leading consulting and IT firms in the past. He is an MBA from S.P. Jain Institute of Management & Research, Mumbai.