Artificial Intelligence, the silver bullet for speed in Clinical Development
April | 2020
Speed in taking much needed vaccines and drugs to market was never as important as now. With the novel Coronavirus (COVID-19) pandemic threatening casualties en-masse, speed of clinical research is a matter of life and death. Quarantine measures, site closures, travel limitations, interruptions to supply chains can hinder research efforts. USFDA has recently recommended sponsors to evaluate alternative methods for clinical assessments such as phone contacts, virtual patient visits, patient treatment adherence and additional safety monitoring1. This indicates that in the digital world, Artificial Intelligence-based clinical development will be the next normal.
Today, cognitive technologies are touching our lives on a day-to-day basis. Almost every industry is striving to implement Artificial Intelligence (AI) to solve business challenges and carry out operational tasks in smarter ways. Life Sciences and Pharmaceutical industry, especially the area of clinical research, is a leading example where humans/scientists are inclined towards intelligent machines/computer programs, and machines are learning rapidly to understand human intelligence.
The recent news of the first AI-generated drug, designed by Exscientia and Sumitomo Dainippon, entering human trials confirms a promising future for AI2.
Shift towards AI
AI is the broader concept, which enables machines to mimic the human brain in performing various tasks, such as, reading, learning and unlearning, memorizing, vision setting, analyzing, planning, communicating, decision making, and problem solving.
Bill Stith, Senior Vice President and Global Head of Health Business at Wipro says, “Automatic machine-driven cognitive functions and mind processes have become a boon to the clinical research industry as they can helps scientists to handle large chunks of information and carry out time-consuming repetitive tasks by replicating human brain's perception and unconscious decision-making ability.”
AI-led advances can enable the clinical development industry to transform their business by reducing complexity, gaining quick insights, saving time and cost of development, and increasing consumer engagement with better outcomes.
AI has become a much-needed building block in the clinical research industry and this paper provides an overview of how AI is influencing Clinical Development.
The potential of AI in clinical development
Global biopharmaceutical R&D market is expected to grow more than USD 1.12 Trillion and Global R&D over USD182 Billion by 20223. It takes on average 10–15 years and more than USD 2.6 Billion to bring a new drug to the market. Approximately half of the time and R&D investment is consumed during clinical development and remaining half in discovery, research and regulatory processes. Although there has been significant focus and increased investments in R&D, approximately 92% clinical trial failure rate has been observed4. Phase III trials consume most of the trial budget and the resulting loss significantly affect total R&D investment.
Success of any clinical development lies in right identification of targets and drugs, effective clinical trial designs with efficient endpoint assessment gears, right patient and investigator selection, and continuous patient monitoring.
AI technologies hold a great potential for improved outcomes, efficiency and effectiveness of clinical development by emphasizing fusion of physical, machine, digital and clinical worlds for safe and rapid generation and dissemination of data for patient wellbeing, Quality of Life (QoL) and optimal management of illness in a targeted population.
As per Gartner survey “2019 CIO Agenda: Life Science Industry Insights”, 34% of industry CIOs call AI a “top technology game changer”. AI tops the list of “game changers” over other technologies in Life Sciences (LS) and all other industries. It is estimated that nearly 50% of 2018-19 AI-related spend supported decision and process augmentation, and a significant uptick in AI related conversations with clients, specifically into LS clinical trial innovation, shows the potential value of AI5. Different AI methods perceived in Clinical Development are depicted in figure 1.
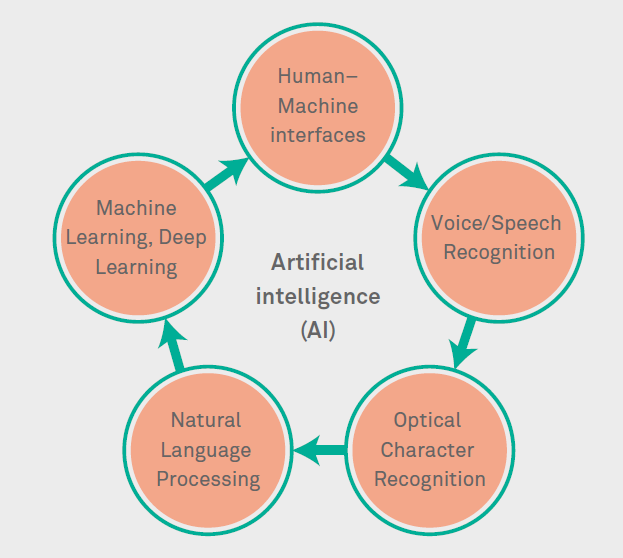
Figure 1: AI methods used in Clinical Development
Transforming clinical development with AI
AI systems assemble statistical algorithms that process huge volumes of input data and generate useful insights and predictions. All AI techniques are self-learning, simulate human intelligence with machines, and can be extensively used for the planning, scheduling and optimization activities in Clinical Development.
Figure 2 summarizes uses of AI in clinical development.
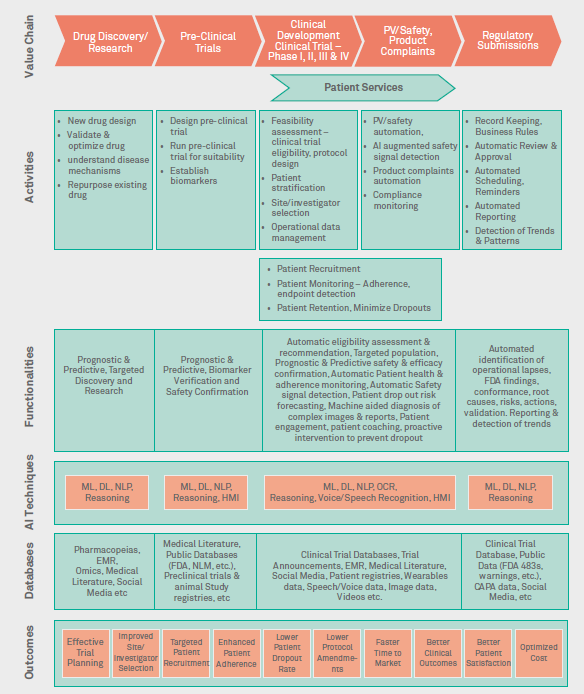
Figure 2: The use of AI in Clinical Development
Drug discovery/research
AI can be used for target identification, in-silico drug design, validation and optimization of drug candidate, and repurposing of existing drugs. NLP-based literature search and ML algorithms can help explore favorable pharmacology profiles and predict physiochemical and pharmacological properties of drugs. This will assist in identifying novel biomarkers, and understanding therapeutic efficacy on multiple disease pathways by screening thousands of potential compound libraries. Predictive analytics and ML can be used to identify new indications for known drugs and match existing drugs with rare diseases.
Pre-clinical trials
AI helps researchers discover insights on cell interactions from large datasets through automatic selection, manipulation and analysis of cells for target validation, toxicology and phenotypic screening to evaluate drug safety and effectiveness. This can help optimize time, cost and uncertainty in planning experiments.
Clinical trials
NLP and ML can be used on Real World Data to enable patient-trial matching by identifying predictive biomarkers to select patients with high probability of treatment outcomes and prioritize prognostic biomarkers to identify disease drivers. Also, these techniques help in measuring drug responses, and designing protocols.
Predictive analytics and ML help screen sites and investigators, predict site performance, and enable centralized risk-based trial monitoring. It can predict and tailor recruitment strategy before launch of clinical trials and assist in dropout risks forecasting and intervention. AI and ML can help process and classify Trial Master File documents providing algorithm-based ability to normalize and validate data.
Chat bots can help sites with initial logical questionnaire-based assessment of patients for Patient Recruitment / Pre-Screening. Site selection can be expedited with chat bots to gather site information like staff qualifications, past performance etc. Chat bots can also collect participants’ data remotely without visiting site directly on eCRFs. Patient diaries and surveys can be replaced with chat bots for electronic patient-reported outcome (ePRO) and electronic clinical outcome assessment (eCOA). Also, chat bots can be explored for eConsent by providing patients clear and easy-to-understand clinical trial information to make informed decisions.
Thus, building knowledge-based AI ecosystem to accelerate operational data management and clinical development can enable site-less/virtual trials.
Patient services
NLP and ML can help analyze medical records to identify and select patients for clinical trials to accelerate 'right' patient recruitment to complete clinical trials faster. Predictive analytics can be used to monitor patient’s health and analyze patient’s behavior and to educate them on use of treatment, thereby improving medication adherence during trials. AI can be used for clinical endpoint detection by assessing potential impact of treatment on the disease by specific symptom/s by analyzing various forms of databases including speech samples, image data etc. Predictive analytics can be used to determine patient dropout risks, potential non-compliant patient and subsequent interventions needed. Chat bots can be used for patient engagement, personal coaching, also by sending automatic personalized messages and notifications to encourage them to continue participating in the trial.
Pharmacovigilance /safety, product complaints
NLP can automatically extract adverse event and product complaints data from various sources such as social media and publications to generate critical insights and to detect safety signals. AI can help simplify Adverse Events, product complaints process by replacing internal monitoring and tracking activities with intelligent risk management, signal detection, aggregate reporting and single case processing. ML can be used to automatically code Adverse Events, improving coding accuracy and speed. This will accelerate safety operations and provide higher quality data and improved compliance. This helps companies refocus time and resources on more strategic endeavors and improve patient safety.
Regulatory submissions
NLP, ML, and predictive analytics can improve efficiency, compliance and transparency for research sites and sponsors, by helping researchers monitor performance and risk indicators for audits, change controls, non-conformance and complaints. AI can automate identification of operational lapses, FDA findings, root causes, risks, reminders, schedulers, actions and validation, automated review and approval, reporting and detection of trends and patterns. This can significantly lower trial cost, time and risk to deliver milestones on time.
AI can help bring products faster to the market and reduce cost of development by leveraging multiple evolving datasets for better insights and decision-making, and to enhance speed, operational efficiency, accuracy and compliance.
The way forward
Although, Clinical Research industry has started experimenting with AI, there have been challenges in adoption of AI including algorithm transparency, data privacy, data integrity, data security and data interoperability issues. Validation of algorithms used for clinical development is vital to understand AI-powered conclusions. Use of patient data is highly sensitive and requires appropriate regulatory compliance measures to protect data privacy. Data integrity and interoperability controls should be in place for data exchange. Data security measures including authorization and authentication are essential. Failure to meet compliance requirements may lead to reputational and financial consequences for manufacturers.
FDA is evolving a strategy to regulate the use of AI in clinical development, emphasizing the use of AI as a confirmatory tool. AI will lead this era as most manufacturers invest in AI to reap benefits of optimized time and cost, leading to faster time to market and competitive advantage.
References
Dr. Sarika Vanarse
Managing Consultant - Life Sciences Consulting Practice, Wipro. Dr. Sarika has over 20 years of cross-functional experience in both technology and business domain for digital strategy, innovation consulting, business transformation, process design, analysis and optimization. She has successfully executed global business transformation programs leveraging cognitive data-driven technologies to accelerate business growth and competitive edge.
For more information, please write to us at health.sbu@wipro.com