Label and artwork management in medical device companies are still seen as an afterthought and therefore, there is not enough concentration as well as investment in this area. However, with new updates such as traceability (UDI) and e-labeling, companies are compelled to invest time and money to be compliant on the regulations required.
One such update is the e-labeling requirement. Health authorities are ensuring e-labeling adherence from marketing authorization holders for their region. For instance, all medical device manufacturers in the European Union have to publish the user information, which is available in labels, technical manuals and instructions of use, on the manufacturer’s website. This is in requirement with the Section 23.1 of Annex I (Chapter III) of the MDR (EU) 745/2017 and section 20.1 of Annex I (Chapter III) of In-vitro Diagnostic MDR (EU) 746/2017. This states that information related to the device needs to be published in electronic form.
There is still heavy dependency on paper-based instruction manuals provided along with the device. Hence, regulatory bodies are ensuring that information pertaining to use of the device is available anywhere and everywhere with the implementation of e-labeling. There may be many reasons with printed copies fading with time, availability of hard copies at the time of use, storing capabilities of the user, etc., implying that paper-based systems are resource intensive. In addition, regulatory bodies want to reduce the carbon footprint which will help companies to reduce waste and country-based printing cost.
E-labeling is gaining favor with the regulatory bodies and emphasizing on publishing electronic content. All Instructions for Use (IFUs) and Direction for Use (DFUs) are required to be available in electronic forms and all device manufactures must comply with the regulations. All major companies have already started working towards this requirement.
What are the current challenges?
What is required for e-labeling?
This requirement will be a part of supply chain requirement, and the right partner can assist you qualitatively and quantitatively and at the right cost, without impacting the current operations.
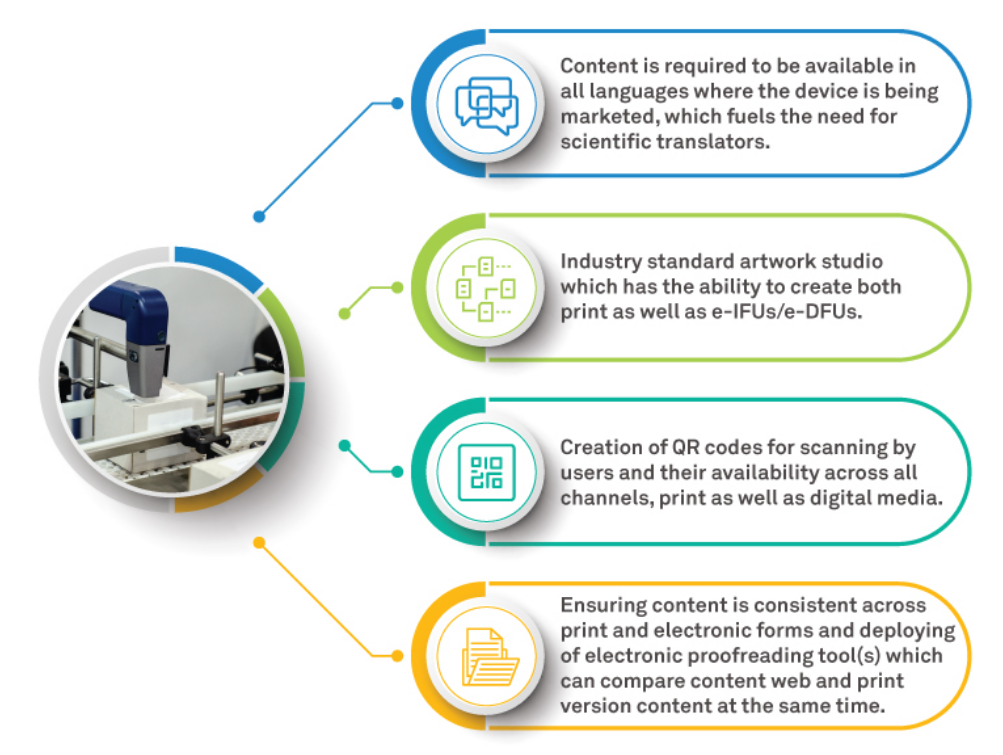
Key criteria for the right partner for e-labeling includes:
Industry :

Pranab Gogoi
Pranab’s experience spans over 21 years across Labeling Artwork Lifecycle Management in the pharmaceutical and life sciences industries regulatory domain. He has worked for major life sciences consulting firms and has catered to customers such as Novartis, GSK, and Apotex as a regulatory SME. He has also successfully transitioned pharmaceutical regulatory processes and projects, implemented related technology and established quality management including audit preparedness and SOP writing.