December | 2019
Blockchain Pharmacovigilance (PV) deals with detection, medical assessments, evaluations, monitoring and, prevention of (ADRs)1. However, the apparent increase in the data volumes of ADRs are putting a lot of pressure on pharmaceutical companies to ensure patient safety, and assure compliance. According to FDA (US Food and Drug Administration), as many as 334,395 new ADR reports were received in quarter 1, 2019 alone2.
This POV will explore how Blockchain can help addressing all the challenges that the pharmaceutical companies face during PV process.
PV plays a critical role in the following areas:
What are the current challenges in Pharmacovigilance?
With the rapid increase of information across the globe, Pharmacovigilance (PV) practices/ departments need to face enormous and endless challenges in keeping up with the public expectation of drug safety, managing various channels/systems to report ADRs and clinical trials, dealing with a complex and large landscape of legacy systems, data security & privacy, and evolving regulations.
It is of utmost importance to have a comprehensive and thorough understanding of current challenges in PV practice to be able to address these challenges and craft an approach using enhanced technological solutions.
PV lifecycle diagram (Figure 1) depicts the overall PV process and the current challenges faced by PV departments.
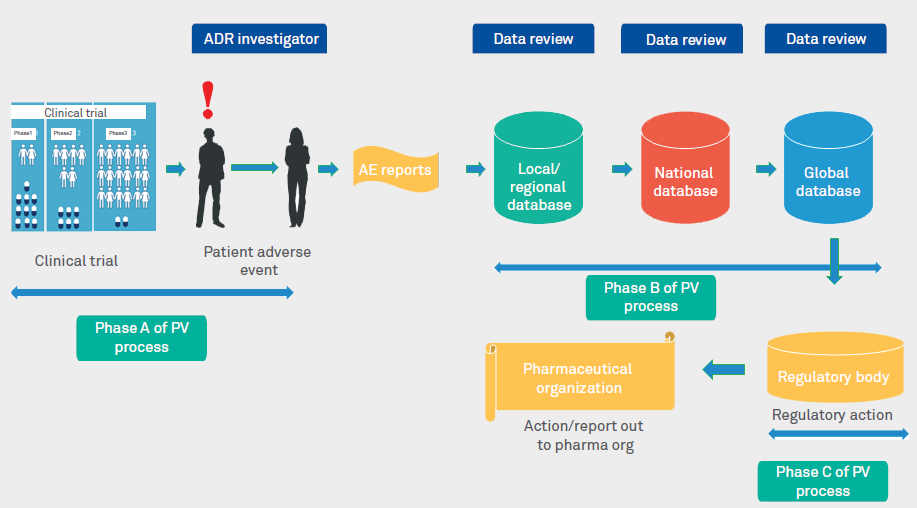
Figure 1: PV lifecycle
Phase A of PV process
Phase B of PV process
Phase C of PV process
How Blockchain can address current challenges in Pharmacovigilance
Due to above said challenges faced in PV process, all pharma and drug companies are exploring Blockchain-based PV solutions to ensure drug monitoring and safety. As per a report, “The healthcare industry is aiming for the most aggressive deployments of Blockchain, with 35% of health and life sciences companies planning to be implement by 2019’’4.
Blockchain provides de-centralized and evenly distributed database in terms of nodes, safeguarding security and transparency of the data. Blockchain allows various entities to process data via various nodes with no central authority. This helps the entities to see real time transactions/revisions without the intervention of any third party. All the relevant parties are connected through nodes via distributed databases, and there is no centralized authority (Figure 2).
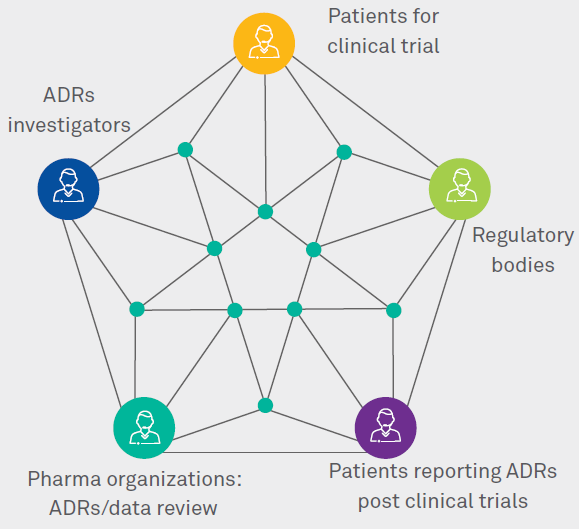
Figure 2: Blockchain addressing challenges faced in PV process
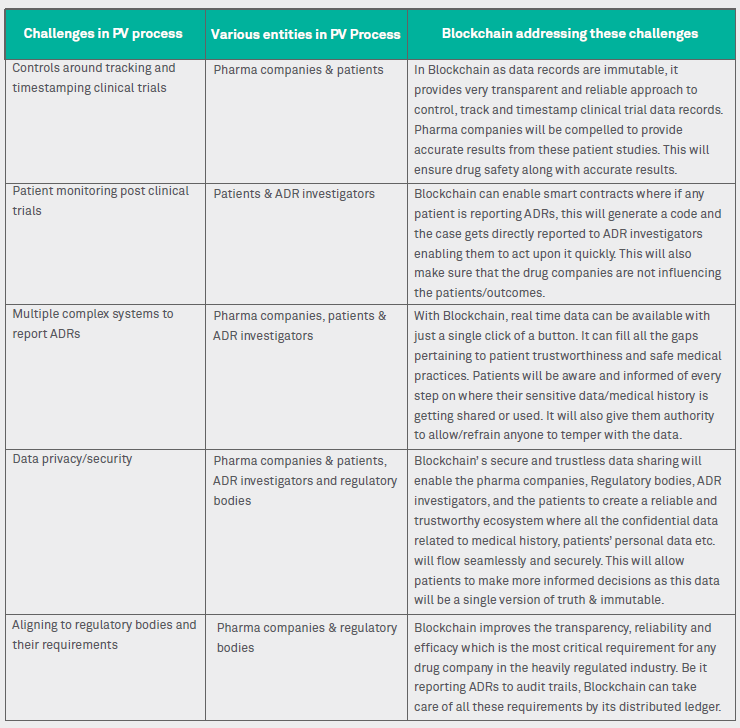
Figure 3: Blockchain addressing challenges in PV process
Are Drug companies using Blockchain?
Conclusion
Blockchain offers significant opportunities to reinvent the way drug companies’ access, collect, distribute, share, leverage, monitor and audit clinical trial data or medical/patient records. However, in order to reap the benefits of Blockchain, drug and pharma companies must overcome barriers to adoption. To facilitate adoption, Pharmacovigilance seems to be a clear use case for the technology.
Blockchain can help address the challenges in Pharmacovigilance (PV). Blockchain provides de-centralized and evenly distributed database in terms of nodes, safeguarding security and transparency of the data.
Blockchain allows various entities to process data via various nodes with no central authority. This helps the entities to see real time transactions/ revisions without the intervention of any third party.
Blockchain offers significant opportunities to reinvent the way drug companies’ access, collect, distribute, share, leverage, monitor and audit clinical trial data or medical/patient records. However, in order to reap the benefits of Blockchain, drug and pharma companies must overcome barriers to adoption. To facilitate adoption, Pharmacovigilance seems to be a clear use case for the technology.
References
Hina Jatale
Digital Operations & Platforms, Enterprise Operations Transformation, Wipro
Hina has been with Wipro for five years and has around ten years of total experience in IT and technology consulting. She has been involved in multiple technology and transformation assignments for CXO level clients across multiple industries spanning various domains, such as MDPS, Manufacturing and Hi-Tech, Consumer.