Blockchain: A Future- Proof Enabler of the Evolving Life Sciences Industry Paradigm
The life sciences industry is in the middle of a profound transformation driven by stressors such as anemic pipelines, shrinking R&D investments, empowered patients, stricter global regulatory frameworks, low margins rise of generics and biosimilars. Companies are struggling to adapt to this new normal and embrace new patient-centric business models driven by real-world evidence.
The new evolving industry paradigm encompasses solutions to the above-mentioned structural problems and new business models that are now possible due to the advances in technology, genomics and medicine.
This document focuses on exploring how blockchain technology can enable the new industry paradigm with its inherent constructs of immutability, disintermediation, transparency, provenance and security.
The new life sciences paradigm
The new industry paradigm is a locus shift from product centricity to patient centricity. The ecosystem is now being shaped around the patient, caregivers and providers, with players exerting increased scrutiny on the cost vs. benefit of drugs and devices.
One common underlying foundation enabling Patient centricity, value-based care, evidence-based medicine, connected health and precision medicine is patient and real world data
Global regulators are responding to growing calls for patient safety and industry accountability by creating new regulatory frameworks and strengthening existing ones. FDA’s Drug Supply Chain Security Act (DSCSA), EMA’s Identification of Medicinal Products (IDMP), Falsified Medicines Directive (FMD) and Good Distribution Practices (GDP) are cases in point of increased regulatory rigor.
New business models such as evidence-based therapies, value-based care, outcome-based pricing, personalized medicine, open innovation and crowdsourced clinical trials will be dominant sustainable models.
A common underlying foundation for all the above mentioned new business models is secure collaboration on patient and research data. With advances in genomics and open source tools such as CRISPR, access to large datasets is imperative for any successful large molecule based innovative product.
The ubiquitous data challenge
The new industry paradigm is a locus shift from product centricity to patient centricity. The ecosystem is now being shaped around the patient, caregivers and providers with payers exerting increased scrutiny on the cost vs. benefit of drugs and devices.
Researchers, patients and volunteers are restricted from sharing and donating their data freely. The root of this problem may lie in the common lack of trust around the usage, security and control of such data.
While data democratization is on the rise, acquiring and securing massive amounts of relevant patient, genomic and research data remains a herculean challenge. The distrust around security, privacy and usage of data is rooted in some of the structural challenges discussed below.

Blockchain technology can enable the creation of secure collaborative platforms where the data is immutable, transparent, secure and auditable for provenance in real time.
What is blockchain?
Blockchain is a shared book of records (ledger) which allows multiple parties (participants) to record information (digital assets) and control how the information is added onto the ledger. Blockchain enables “value exchange” – be it for currency, patient health records, documentation or any type of ownership of assets. At the heart of it, blockchain allows peer-to-peer exchange in a multi-party ecosystem[1].
“At its core, Blockchain is nothing more than a special kind of distributed database. This special kind of distributed database features an ecosystem of selected participants, which could include anything from upstream suppliers to downstream consumers, regulators, or other appropriate stakeholders.” - Biran Behelendrof (Executive Director of Hyperledger Project)
Each member (called node) of the ecosystem holds a copy of a distributed electronic ledger. These ledgers get periodically synced up when new transactions are validated and agreed upon by a majority of the members of the network. This helps in checking against any accidental or malicious data inflow, which can be detrimental to the entire network. This creates a network of records that is nearly impervious to alteration or fraud. Moreover, since each participant is using an identical record, there is no need for any reconciliation. This reduces costs and effort, and accelerates settlement time.
Blockchain technology works on four basic tenets [1]:
Key characteristics of blockchain
The new industry paradigm is a locus shift from product centricity to patient centricity. The ecosystem is now being shaped around the patient, caregivers and providers with payers exerting increased scrutiny on the cost vs. benefit of drugs and devices.
Type of blockchain networks
There are typically three types of blockchain networks:
It costs between $100M to $ 2B over 10 years to bring a novel product to market through extensive clinical trials. It is imperative that the content/data each player brings to the table and generates as part of the study, is tamper proof.
Blockchain in life sciences
Research and Development (R&D)
With the advent of open innovation, crowdsourced clinical trials, democratization of data and shrinking R&D funds, companies and researchers are collaborating across the ecosystem including competitors to co-innovate.
Companies are also waiting longer into the research projects for solid evidence before filing for expensive patents.
Blockchain provides the required robust IP protection, transparency and verifiability in real time across globally dispersed teams. As each participant uploads their research data onto the chain, it is recorded as immutable time-stamped transactions. At any point of time, it is easy to trace ownership and provenance of the content or portions of the IP to specific entities or individuals. This will enable open collaboration without the fear of IP theft.
Clinical trials
Clinical trials are complex, long and high risk.
Large global studies with multiple trial arms and investigative sites have several CROs, CMOs, Principal Investigators, Clinical Research Associates and healthcare providers collaborating on a daily basis generating massive amounts of clinical and operational data.
Fraud and data tampering can sometime get buried in the volumes of data-skewing safety and efficacy analytics and can be detrimental to the health of the clinical trial and subjects.
How blockchain can help:
Cold chain monitoring
With the rise of biopharma, cold chain logistics is gaining new prominence and focus. Many biotech drugs require temperature-controlled shipping and storage.
Temperature excursions continue to be a major challenge and result in about 20% of spoilage and product wastage. This can cause harm to patients and brief drug shortages where required. A similar scenario exists for drugs that are sensitive to light, humidity, vibration, electromagnetic force etc.
Combining blockchain and IoT technologies, it is possible to measure temperature at a unit of sale level in real time on land, air and sea. This temperature data is uploaded onto blockchain and associated with the batch record of the product.
Any excursion in temperature or any other validated environmental factors are recorded as immutable records and are accessible to all the authorized parties including the patient and regulatory agencies.
The World Health Organization estimates that up to 30% of drugs available in Asia, Africa and Latin America are counterfeits. Average for global counterfeits rises to 10 percent. 100,0000 lives per year are lost just in Africa.
Counterfeit drugs
Counterfeiting is a global problem and it no longer affects just the consumer goods industry. It is unquestionable how lucrative the counterfeit drug business is with hefty margins that exceed drug trafficking and carry comparatively low risk.
How blockchain can help?
Inventory management
Inventory management in life sciences falls under the broader umbrella of Good Practices (GxP). Drug and device manufacturers maintain consignment stocks globally and it is critical, both from a regulatory and financial aspect, to be able to account for that inventory at a unit level. With blockchain, it is possible for all the authorized participants to uniquely identify and track the current location and chain of custody of the product at a granular level.
Product expiration
Expired products can be ineffective and even cause harm to the patients. It is critical to be able to identify and dispose expired products to prevent dumpster-diving and reintroduction into the supply chain. Blockchain can track and provide product expiration alerts to the entity with custody of the product. Further, by using blockchain for reverse logistics, products can be tracked until they are disposed of, according to standard operating procedures.
Product recalls
Product recalls are expensive and time-sensitive, requiring quick location and identification of the affected products. Unfortunately, current systems are inadequate or limited for this functionality. Some of it is due to multiple sales channels involved and others due to supply chain complexities.
Blockchain can readily provide location and chain of custody information at a unit level by a simple user search by product, batch, manufacturer etc.
Participants who had handled the recalled products can also be alerted programmatically when the manufacturer broadcasts unique product identifiers from the recalled batch.
This will help with targeted speedy recall improving patient safety, manufacturer’s reputation and reduce the overall cost of recall.
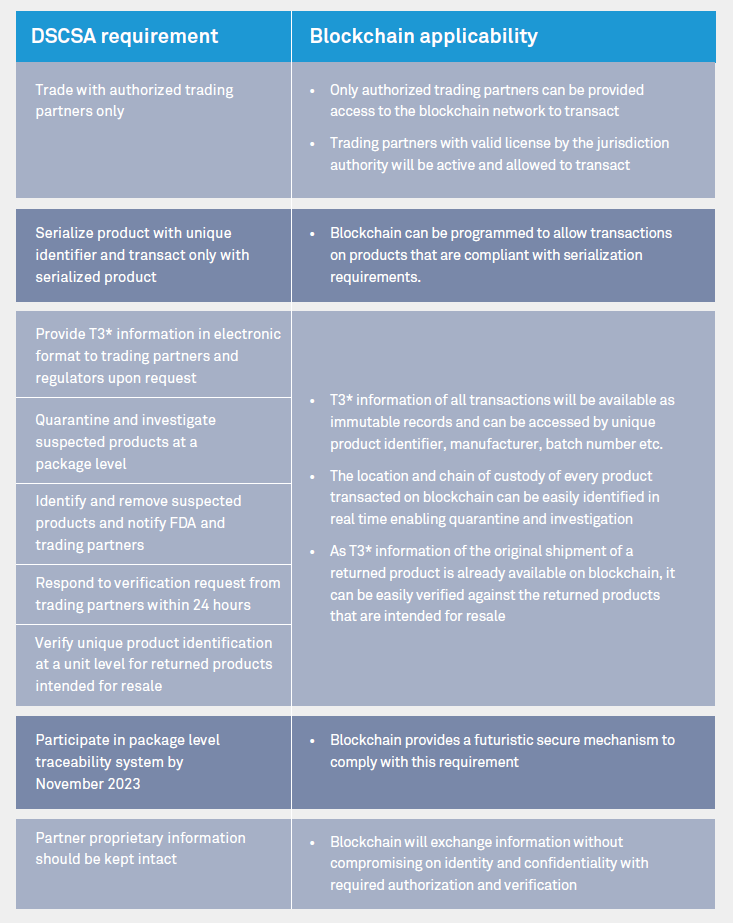
To protect patient safety, regulatory agencies are implementing supply chain security frameworks. Once implemented it will require manufacturers, re-packagers, wholesale distributors and dispensers to comply.
Regulatory compliance requirements
Case studies
Pharma Cold Chain - A Wipro innovation for one of the world’s leading biopharmaceutical companies
A Digital Supply Chain & Anti-Counterfeiting Solution built on IoT, Blockchain and Cloud technologies
The solution involves creating an industry framework that secures the supply chain and prevents cold chain wastage by generating geospatial alerts in real-time in the event of a temperature excursion or counterfeiting attempt. The data of shipment and any anomalies during the shipment are recorded on a private permissioned blockchain network.
This platform helps in the minimization of drug spoilage during shipment, which in turn results in improving patient safety. It gives an immutable audit trail across the supply chain that helps in identifying any counterfeiting attempts, thereby reducing losses for the organization from the sale of counterfeit products.
Pharmaceutical Track and Trace - A Wipro solution developed for one of the world’s leading pharmaceutical companies
A pharma track and trace solution built on IoT and blockchain technologies
Our Proof of Concept (PoC) brings the pharmaceutical company, distributors, customs, wholesalers and retailers on a common blockchain platform to track and trace the provenance of international drug shipments. Another key aspect of this project is to ensure that all compliance documentation required at each stage of the supply chain is made available proactively through blockchain.
The solution provides real-time visibility into the status of pharmaceutical drugs shipments, better inventory reconciliation, on-demand document & compliance check, and improved collaboration amongst multiple parties.
Lab to Launch - A Wipro solution developed for one of the world’s leading pharmaceutical companies
A blockchain-based solution enabling collaboration across global teams in a pharmaceutical drug launch lifecycle
A new drug launch consists of multiple, complex and interdependent processes within an organization. These processes include government approvals, internal corporate approvals, ground team mobilization as well as the entire supply chain enablement. This entire process spans over 3-5 years. Our PoC brings together corporate, marketing, finance, internal regulatory teams, medical teams and supply chain teams on a common blockchain collaboration platform. In the overall set of activities, multiple internal and external approvals and certifications are recorded on blockchain.
Speed, accuracy and auditability of information are very critical during these processes. Blockchain enables seamless, transparent and proactive information exchange between all stakeholders.
About blockchain at Wipro
Wipro is a partner of choice for our clients across industries in driving Blockchain innovation. We achieve this through our industry recognized strategic advisory and consulting services combined with a strong portfolio of industry solutions and patents developed in our center of excellence. We constantly invest in creating a strong Blockchain community and developing talent for building deep technology expertise across multiple leading Blockchain platforms. We have a comprehensive Blockchain ecosystem of technology partners, industry and regulatory bodies and academia to deliver value to our clients by driving enterprise-grade production Blockchain networks.
Sujan Thanjavuru,
Head of Strategy & Consulting, Life Sciences
Anirudh Sharma,
Designation: Blockchain Consultant, Blockchain Practice