April | 2020
A consolidated strategy is required to improve manufacturing process efficiency by harmonizing processes for electronic batch records (EBR) and electronic device history records (eDHR) for pharmaceutical and medical devices plants. This can help improve real-time visibility, data integrity and enhance data quality besides ensuring compliance and traceability. This article provides an overview on a case study for electronic batch records and electronic device history records process harmonization strategy.
The life sciences and medical devices industries are currently facing many challenges in a competitive global market. To maintain market position and meet customer demands, the organizations are under pressure to introduce new products early to the market with optimized costs. There is another challenge to conform to evolving local and global regulatory guidelines for ensuring manufacturing quality and minimizing product recall, which can be achieved by keeping track of batch records (BR) and device history records (DHR). Regulatory agencies emphasize keeping track of production details for each product manufactured by organizations, which is a cumbersome process, as management of BR and DHR is largely paper based, people dependent process in many organizations.
The manual processes take a lot of time and efforts and are often error prone, impacting overall costs and operations. Incorrect entries, unclear documents, rounding and calculation errors can significantly affect approval processes. Industry is exploring an efficient process to generate electronic, digitized batch record and device history records.
Business Needs
Data level
Improving manufacturing processes by replacing paper based mechanism with electronic data capture is utmost important.
Typical outcome at data level is:
Plant level
Due to the unique nature of production processes across various manufacturing plants, data capture processes and compliance needs are inherently complex. Examples of these complexities include:
Need of the Hour– Best Practices
EBR/eDHR implementation for batch records and device history records automation has become paramount and this can serve as a springboard for connecting the stakeholders for governance, data capture, and quality as well as providing a platform for manufacturing process improvement.
It is recommended that process mapping should be performed to identify and eliminate waste before the EBR implementation.
Industries are adopting some of the best practices to move towards digitization and automation of EBR and eDHR, which are discussed below.
Elements of Electronic Batch Records
Key enablers of EBR/eDHR for each benefit area and specific examples of improvements that will be realized when manufacturing execution and data capture moves from manual/paper based to automated/electronic are depicted in figure 1.
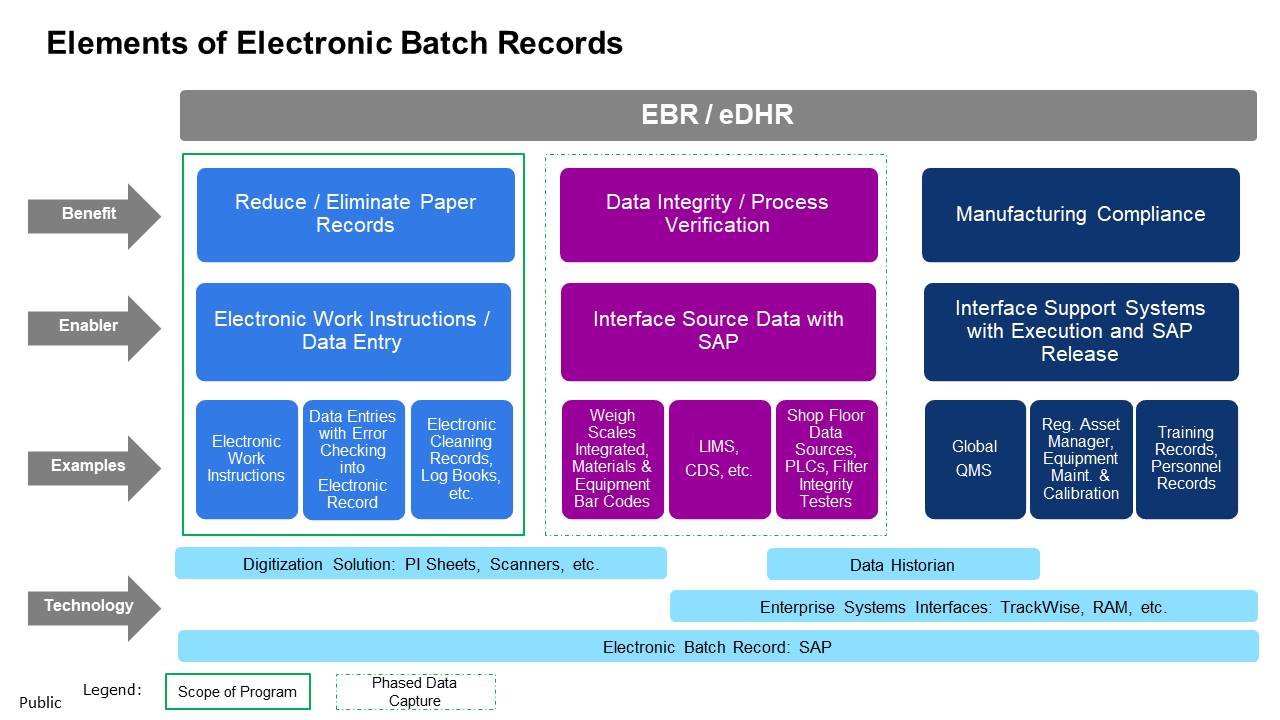
Figure 1: Elements of Electronic Batch Records
The technology layer in the figure 1 provides general descriptions of the types of applications cutting across multiple benefits “pillars” to enable EBR creation.
These descriptions are generic as there could be multiple technical pathways to achieve the endpoint. For instance, the requirements for a digitization solution can be met by applications with custom PI sheets in SAP or electronic work instructions and data capture.
Key Business Benefits
Manufacturing process harmonization and digitization benefits business with:
Digitization of EBR and eDHR coupled with manufacturing process standardization can help improve compliance, data quality and data integrity, and enhance operational excellence and global collaboration.
Dr.Sarika Vanarse
Managing Consultant – Life Sciences & Medical Devices, Wipro Ltd.
Dr. Sarika is a Consulting leader with Wipro’s Health Consulting Life Sciences and Medical Devices practice focusing on driving business and technology transformation programs across value chain.
Dr. Sarika Vanarse can be reached at sarika.vanarse@wipro.com
Dr. Rao Teki
Global Head - R&D Innovation and Transformation, Wipro Ltd.
Dr. Rao is a Consulting Partner and Global Head - R&D Innovation and Transformation focusing on driving innovation and transformation of R&D business.
Dr. Rao Teki can be reached at rao.teki@wipro.com